Shrimply Explained is currently undergoing renovations. Please don't buy anything yet. Thank you!
If you get nothing else out of this lesson, remember that having stable parameters is much more important than getting the “perfect” pH, KH, or GH, even if your tank is slightly outside the recommended ranges. DO NOT chase a specific value.
This lesson provides an overview in clear terms than anyone can understand. Click here if you are interested in a more detailed scientific explanation of pH and KH.
As with all animals, water plays a life-or-death role for your shrimp. This makes understanding key parameters like pH, KH, and GH the most important thing you can do to provide them with a healthy ecosystem. In addition, we cannot talk about aspects of tank setup and maintenance without understanding what these parameters are, why they change, and how to control them. This lesson covers everything you need to know about pH and KH, with information on GH provided in the next lesson.
Shrimply Explained is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com.
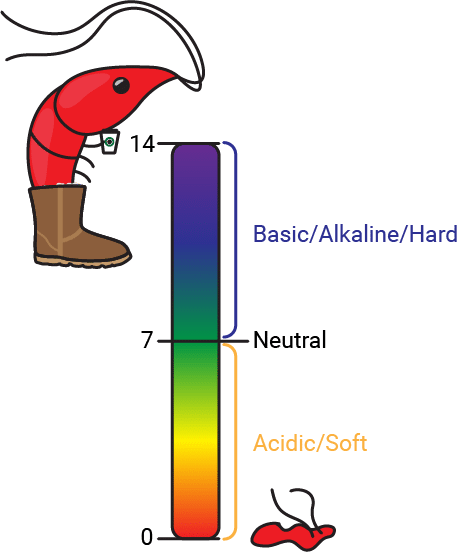
The potential of Hydrogen, or pH, is a measure of the concentration of hydrogen ions (H+) present in your water. As you may remember from nightmares about chemistry class, pH is measured on a scale of 0-14, with below 7 being acidic, at 7 being neutral, and above 7 being basic or alkaline. In terms of hydrogen ion concentration, 14 is the least concentrated and 0 is the most concentrated (although there are superacids much stronger than 0 pH). Acidic water is also referred to as soft water while alkaline water is called hard water.
Now, let's transport you back into another chemistry nightmare that we've all had:
Lightning flashes through the window, illuminating rows of bound books and burbling bottles. A cackle crawls from the shadows and echoes off the stone walls but your captor remains hidden. You dangle from a sinister pulley system, tied up and slowly dropping toward a steaming cauldron where your flesh and bones will disintegrate in acid. Is this how it ends??
Now, let's pause there and consider - Why does acid dissolve your body--or anything else?
The reason that acids (and bases) can melt through flesh is because they are extremely reactive. Your flesh starts turning into different compounds that are not compatible with the rest of your body... and you know the rest. Now, obviously, there should be nothing so extreme happening in your tank but the point is that many compounds in the water—and in the body of your shrimp—change as pH varies. Let’s look at ammonia (NH4+) as an example.
Ammonia can be found in two forms in your tank depending on the pH. At 7 pH or below, 99% of all ammonia is in a relatively non-toxic form. As pH increases, the percentage found in the toxic form increases (approximately 5% taking on a toxic form at 8 pH, 10% at 8.4 pH, and 25% at 8.8 pH). So while you may not notice anything wrong if your tank has low pH, a sudden increase in pH from a water change or chemical additive may create toxic ammonia that harms your shrimp. That is part of why it is so important to make changes to the tank slowly.
For those interested in a slightly more scientific explanation, the non-toxic form is ammonium (NH4+), which occurs due to the higher concentration of H+ ions available in acidic conditions. This hydrogen is more likely to be lost in basic conditions, forming the toxic ammonia (NH3).
Shrimp are extremely sensitive to these changes so their bodies experience stress. This makes them more susceptible to diseases, if the change doesn't kill them outright. Extreme pH shifts are a common cause of fish and shrimp death, but even stable pH outside of each species limits causes problems, including but not limited to: stunted and slow growth, low reproduction rates, molting problems, diseases, loss of eggs, death, etc. As such, understanding why pH changes and how to control it are very important to your success as a shrimp keeper.

pH change is caused by a wide variety of sources but the two that are consistent across all tanks--and all water sources in the world--are natural and necessary cycles. First is the nitrogen cycle, which you may be familiar with if you’ve had an aquarium before. This process is responsible for converting waste (ammonia from animals, leftover food, dead or dying organisms) into less harmful chemicals.
The nitrogen cycle may be disrupted due to excess ammonia or bacteria die off, leading to significant pH changes. Adding live, fast-growing plants is a great way to avoid these sudden pH changes because plants absorb ammonia before excess gets converted by the nitrogen cycle (dropping pH) or built up if the cycle is overwhelmed (raising pH).
Second is the photosynthesis cycle. It is not responsible for fast pH changes but instead causes a slow increase in pH throughout the day and a decrease at night. Normally, the rate of change is so slow that shrimp are not affected but it can become a problem if the pH varies too far out of normal ranges.
Side note: When tracking pH in your tank, it is always best to measure at the same time each day due to the photosynthesis cycle.
1. Test Strips
These are cheap paper strips that change color after being dipped into water. Strips are generally the least accurate method because different amounts of water tend to stay on when the strip is pulled out of water. The colors also tend to run if not kept flat which can be annoying. Regardless, they are a quick way to get an estimate and ensure nothing is seriously wrong with your tank. We have found these test strips from Amazon work relatively well as a cheap way of measuring pH (and every other important water parameter):
2. Liquid Test Kits
The most common option is using a liquid test kit such as the API Freshwater Master Test Kit. While more expensive, liquid test kits are more accurate and easier to interpret. In addition to pH, the Master Kit also includes tests for ammonia, nitrites, and nitrates so is a great deal to track the long-term health of your tank.
2. Electronic pH Meter
Lastly is an electronic pH meter, which provides a digital readout when placed in tank water. It is the fastest and most accurate option, while still being reasonably priced. The only issue is that pH meters must be calibrated to ensure accuracy, which can be a hassle.
pH is closely tied to KH in your tank so control methods are the same. As such, these methods are combined into one section at the end of this lesson.
If already familiar with what KH is and why it matters for your shrimp, then feel free to skip to Controlling pH and KH.
KH stands for carbonate hardness (Karbonate in german) but may also be called total alkalinity. The units of measurement are:
If you ever need to convert between the two, then know that 1 dKH is equivalent to 17.9 ppm (rounding up to 20 may make it easier to remember).
Higher KH causes higher pH and resists pH fluctuations—an effect known as buffering--while lower KH does the opposite. The analogy below may be helpful to understand how buffering works:
For anyone not familiar with bowling, the goal is to roll a ball down the lane (a narrow strip of smooth floor) to knock over as many pins as possible. On either side of the lane are gutters that the ball may fall into, directing the ball to the side of the pins and preventing you from getting points. For beginning bowlers, bowling alleys often block the gutters using bumpers, which makes it easier to hit the pins and score.
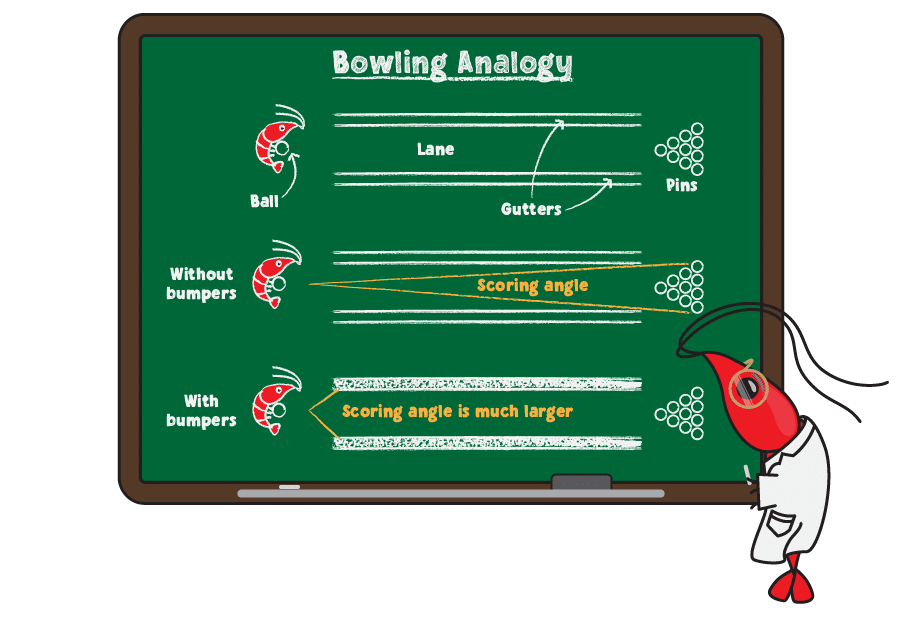
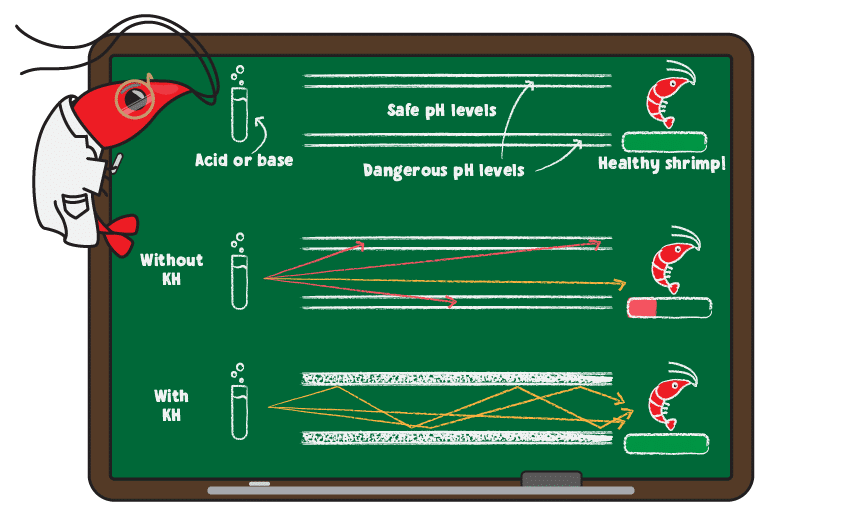
Think of KH like bumpers while bowling. Without bumpers (KH), there is a narrow range of throws that allow the ball to score (get stable pH in proper range) without going into the gutter. When you add bumpers (good KH levels), then you have a much better chance of scoring (getting the right pH in your tank).
Side note: The word "bumper" is very close to "buffer", which may help you remember the role of KH.
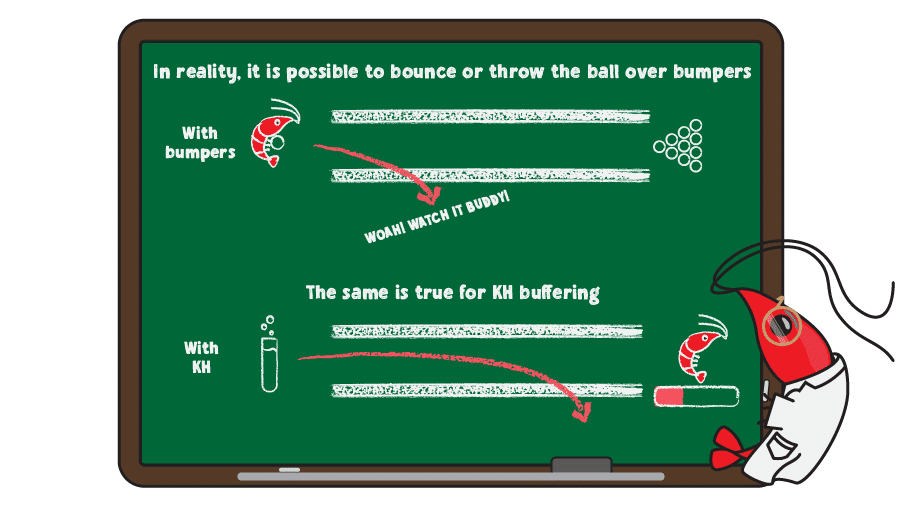
Even bumpers won’t allow every ball to score points , as balls may bounce or be thrown over bumpers. This is true of KH, as well, since too much of an acid or base overwhelms the buffering system, resulting in rapid pH change.
To make even the strongest, most uncoordinated person score in bowling, you could encase the entire lane in a cage of bumpers so the ball can be thrown in any direction and still hit the pins. Needless to say, that would be expensive and impractical for most bowling alleys and bowlers. There is such a thing as too many bumpers.
The same is true of KH because, while buffering is great for preventing pH swings, too much KH causes high pH that is stressful for your shrimp.
KH decreases over time because it gets used up in the nitrogen cycle. In addition, sources of acid (such as active substrates used in planted shrimp tanks) also reduce KH. Water changes may replenish KH but there are other methods of control covered in later sections.

Tired of problematic parameters, mysterious shrimp deaths, and conflicting advice? The Science of Shrimp Keeping cuts through the confusion with 300+ pages of Shrimply Explained, science-backed solutions—so you can finally keep thriving shrimp with confidence.


1. Reverse osmosis, deionized, or distilled water (AKA RO/DI water)
This water is found at most grocery or fish stores and has been purified to remove almost all salts and minerals from it. As a result, the pH is just below 7 and dKH is almost 0. Shrimp keepers that have alkaline tap water may mix in some RO/DI when setting up their tank and performing water changes. This lowers the pH and KH depending on how much the tap water is diluted. For example, if you have 12 dKH tap water and want the water added to your tank to have 4 dKH, then you need to mix 1/3 tap water with 2/3 RO/DI water.
Be aware that RO/DI water has approximately 0 dGH so affects GH, as well. More on that in the next lesson.
2. Buffering Substrate
Buffering substrate like ADA Aquasoil Amazonia and Fluval Stratum are acidic soils, meaning they release hydrogen ions into the water. This counteracts any basic/alkaline compounds in the water--including carbonate--which decreases pH and KH. These products typically lower and maintain the pH below 7. They do not affect GH.
Similar to KH, these substrates lose their buffering ability over time. As a result, they must be replaced periodically (every 6-24 months) to keep pH and KH low. Their buffering ability is also depleted faster if they are added to high pH/KH water, which is why soft water is recommended with these substrates. To avoid rescaping your entire tank each time new substrate needs to be added, you may put the substrate in an easy-to-remove container. This is not a very aesthetically pleasing solution, however.
Due to how expensive buffering substrates are and what a hassle they are to replace, we mainly recommend using them when keeping caridina shrimp, as they require low pH and KH. These substrates also release ammonia initially so be careful when adding them to tanks that are already established.
Click on the products below if you are interested in learning more or purchasing them from Amazon:
3. Driftwood, Peat Moss, Chola Wood and Indian Almond Leaves (catappa leaves)
These natural materials all leach acidic compounds called tannins, which reduce KH and pH. The amount they reduce pH and KH depends on how many tannins are available in these natural materials, which may vary by batch. This can sometimes result in very little effect overall so this method is not super controlled. Peat moss and Indian almond leaves also lower GH (more on that in the next section).
In addition to reducing pH, there is some scientific evidence that tannins help protect your aquatic friends from diseases and improve healing after injuries. Dealing with shrimp disease is never fun, which is why we recommend having at least one item that releases tannins in your tank at all times.
One thing to be aware of is that tannins tint your water slightly brown. A lot of tannins in the water create a blackwater environment that is desired by certain breeds of fish. If you do not like the aesthetics of blackwater, then the wood/moss/leaves may be soaked in boiling water before adding them. Be aware that this results in fewer tannins released into your tank, thereby reducing the impact on pH, KH, and shrimp health.
Click on the products below if you are interested in learning more or purchasing them from Amazon:
Side Note: Peat Moss NOT Spaghnum Moss
While spaghnum moss and peat moss come from the same plant, they are not interchangeable. They have different harvesting methods that result in very different effects on water quality when added to your tank. Spaghnum moss is harvested directly from the living plant before being dried and used to maintain a moist environment for house or garden plants. In contrast, peat moss dies, sinks to the bottom of a bog in Canada, then gets mixed in with a bunch of other dead plant and animal matter before being harvested and dried. As a result, peat moss is far more acidic and provides additional nutrients to the water while spaghnum moss is of neutral pH and has no benefit to an aquarium (although it can be great for paludariums and terrariums). If you’re ever confused, just remember this rhyme:
Peat is neat.
Spaghnum is dumb.
Is it an unnecessarily aggressive saying toward regular spaghnum moss? Possibly - but that just makes it easier to remember.
4. Water Changes
If your local water is softer than your tank water, then adding dechlorinated tap water to your tank is likely the cheapest way of lowering pH and KH. Keep in mind that, if you don’t normally use tap water for water changes, then you may introduce unwanted nitrates or metals (from rusted pipes) into your tank.
5. Acid buffers
Acid buffers are an option to lower pH and KH but are generally not something we recommend for two reasons:
If done right, acid buffers are effective at lowering KH, but we prefer to use the other methods mentioned above because they have a more gradual effect on the water so mistakes are less dangerous.
1. Baking Soda
Baking soda is food-grade sodium bicarbonate. It is very safe, can be added in a controlled way, and is very cheap, making it the best option to modify pH and KH. 1/4 teaspoon (approximately 1.25 grams) of baking soda raises KH in a 10g (38L) tank by approximately 1 dKH. When adding it, we recommend mixing it in water outside of the tank until dissolved, then adding the water back in slowly - ideally dripping it in over an hour or so to avoid sudden pH changes.
2. Crushed Coral/Aragonite, or Wonder Shells
These products are made of calcium carbonate and dissolve slowly in water to raise pH and KH (and GH). Each one may be added to the substrate or filter, but we recommend starting with a mesh bag in the filter so it is easily removable if you notice the pH rising too quickly. Wonder Shells do not need to be kept in a mesh bag.
For the sake of safety, it is also best to start with a small amount, monitor parameters closely for a few days, then add or remove some after seeing the effects.
3. Water Changes
If your local water has higher pH and KH than your tank, then adding dechlorinated tap water may be a cheap solution. As mentioned in Lowering pH and KH, this may introduce unwanted nitrates or metals (from rusted pipes) into your tank if you don’t normally use tap water.
4. Alkaline buffers
We do not recommend these products in your shrimp tank. Many of these products are basically just baking soda, except they may get around food safety standards and potentially be lower quality (less safe) than baking soda.
Whew, that was a lot to go over. Now, you should have a good grasp on what pH and KH are, what role they play in your aquarium, how to measure them, and how to control them. With all that under your belt, you are one step closer to becoming a shrimp keeping master.
If you still have questions, please reach out on social media or send us an email at contact@shrimplyexplained.com. We are here to help however we can and love meeting new members of the community!
Click on Next Lesson to be taken to The Basics of GH for Shrimp Tanks and ensure a successful shrimp keeping journey.